Uncovering insights hidden within the psoriasis transcriptome.
Herein we explain how our novel data mining strategies for RNA-Seq datasets lead to new insights into the pathophysiology of psoraisis, an immune-mediated disease involving the skin. This story can also be found in our recently published manuscript in JCI Insight. You can click here to read the entire publication (subscription NOT required).Introduction
The skin transcriptome is the sum of all messenger RNA molecules expressed in the skin. About a decade ago, next generation DNA sequencing technologies allowed researchers to investigate the skin transcriptome in the setting of psoriasis. This led to the discovery of thousands of genes that were differentially expressed in psoriasis skin; But are all of these genes involved in psoriasis pathophysiology? As is the case with other diseases, knowing the sum of all expressed genes in psoriasis is only a small piece of the puzzle. How to decipher the psoriasis transcriptome has been a challenge that our research team and others are still working on. One strategy to narrow down the thousands of differentially genes to a more reasonable number is to set a threshold as to what represents a meaningful up or down regulation in gene expression. For example, is it meaningful if a gene's expression is increased by only 1.2 fold in the setting of psoriasis?
Meaningfulness is different from significance, which simply means that the observed change is not likely to be due chance. When analyzing DNA sequencing data for significance, p values need to be adjusted for multiple testing, as transcriptome datasets have thousands of genes. Programs used for such tasks take many factors into consideration (number of uniquely mapping reads, total number of genes monitored, variation in read counts etc). However, even when thresholds for significance and meaningfulness are met, investigators can easily be led astray - as data crunching by these classical methods often over or under emphasize the importance of a gene.
The error of meaningfulness thresholds
It is common to set a 2-fold increase or decrease in gene expression as a threshold for meaningfulness. However, is this the best way to identify differentially expressed genes of interest? It depends on the gene, its expression level, and the situation. Lets take for example TNF. During the 1990's, an area of immunology when virtually all cytokines were categorized as belonging to either Th1 or Th2 immune responses, TNF and IFN-g were viewed as the classical Th1 cytokines. This led to the testing of anti-IFN-g and anti-TNF-based therapies in animal models of autoimmunity. In such experiments, anti-TNF therapies showed superior efficacy. In contrast, anti-IFN-g treatments sometimes made autoimmunity paradoxically more severe. Eventually, anti-TNF therapies also proved useful in treating a variety of human autoimmune diseases. With regards to psoriasis, anti-TNF biologic therapies are indeed effective treatments. Although newer psoriasis biologic treatments target different molecules, the prototypic anti-TNF agent, infliximab, is still arguable one of the fastest and most effective medications for psoriasis (in terms of skin clearing at 10 weeks). However, the effectiveness of anti-TNF-based therapies would not have been predicted from TNF gene expression in psoriasis plaques. In comparison to healthy control skin or uninvolved skin, TNF expression within a psoriatic plaque is only marginally increased. In reality, TNF is made by numerous cell types not simply Th1 cells and relatively small changes in its expression have dramatic effects on tissue homeostasis. Thus, TNF blockade has dramatic effects on pathogenic immune responses. Setting a 2-fold change as a threshold for meaningfulness would absolutely miss important genes such as TNF.
For the next example lets consider a hypothetical gene expressed by T cells. If in the setting of psoriasis the expression of this hypothetical gene drops by 30% in T cells, its expression in psoriatic skin will paradoxical not be decreased at all. In fact, its tissue expression will rise more than 2-fold, which may lead some investigators to erroneously conclude that the "up-regulated" gene is involved in the pathophysiology of psoriasis. In this example, the hypothetical gene is decreased in T cells but increased in psoriasis plaques. The increased expression is a result of the simple fact that psoriatic plaques have more T cells not that the expression of the gene is otherwise increased. It is likely that the vast majority of the upregulated genes in psoriasis are a mere consequence of the infiltrating immune cells or proliferation of existing cells within the skin. These are just some examples of why deciphering transcriptomic data is difficult. One method we have chosen to overcome some of these issues is to look at relative gene expression, that is the expression of one gene relative to reference immune genes, especially the genes that encode immune receptors such as the T cell receptor genes. Another strategy is to do single cell sequencing. Both strategies have advantages and disadvantages. For example, the cell preparation techniques used for single cell sequencing will alter the cellular transcriptome.
T cell receptors and T cell repertoire analysis
As just stated, we often use TCR genes as reference genes when characterizing an immune response. However, this strategy has a variety of barriers. TCRs are encoded by productive rearrangements of the alpha beta or gamma delta TCR genes. For the beta and delta chains, the genetic recombination events involve variable (V), diversity (D), joining (J), and constant (C) region exons. This allows for the immune system to express an almost infinite number of immune receptors to project of from invading pathogens and cancer.
Traditionally, TCR genes are sequenced directly from the products of TCR-specific PCR reactions. TCR mining of RNA-Seq datasets is an alternative approach to characterize T cell repertoires, but requires the use of an appropriate bioinformatics alignment pipeline. In our recent JCI Insight manuscript we developed and validated a pipeline to estimate the overall and relative expression of TCR V/J/C segments. Our method has the advantage over traditional TCR repertoire analysis in that it allows investigators to conduct correlative studies between TCR genes and other genes of interest; this is because it detects all TCR-mapping reads in an RNA-Seq dataset. In contrast, bioinformatics tools focused solely on analysis of the TCR CDR3 region cannot accurately estimate TCR gene segment usage and CDR3-mapping only yields a small handful of reads per RNA-Seq dataset, making correlative studies difficult. Also, traditional RNA-Seq analysis pipelines are not aware of TCR rearrangements and may not take into account gene rearrangements, considering them low quality mapping reads.
Our approach allowed us to not only identify novel psoriasis-associated TCR gene segments, but also correlate the transcription of individual alpha beta and gamma delta TCR gene segments with genes of known importance to the pathophysiology of psoriasis. Four independently acquired RNA-Seq sample sets allowed for rigorous statistical validation of our results.
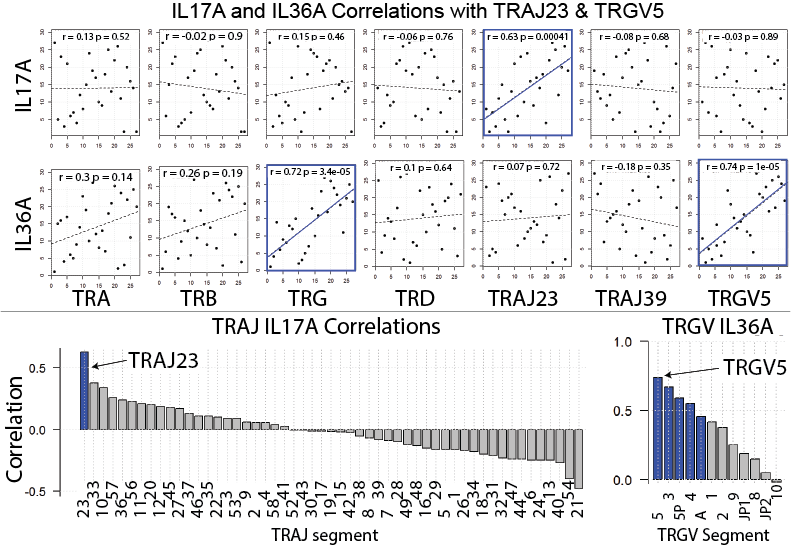
Our manuscript highlights a many exciting findings. For example, by focusing on the expression of individual TCR gene segments relative to all TCR gene segments of the same family, we discovered that TRAJ23 is proportionally over expressed and TRAJ39 is proportionally under expressed in psoriasis (Bonferroni p = 6.19 x 10e-27 and 1.43 x 10e-248, respectively). (TRAJ23 and TRAJ39 are TCR alpha gene segments). We also found that a TCR gamma gene segment is minimally over expressed in psoriasis, TRGV5 (Bonferroni p = 6.03 x 10e-13)). A finding that we were able to validate across 4 different RNA-Seq datasets. The figure below summarizes these results. Please see the full manuscript for more details. This same strategy can be applied to other autoimmune and cancer datasets, and thus carries wide applications. Importantly, our results differ from the alpha beta gene segments previously reported to be disproportionately overrepresented in psoriasis (1-3). These highly conflicting results may be due to the exceedingly small sample sizes used in the prior studies, which for the most part lacked summary statistics to evaluate the significance of the psoriasis-associated TCR genes.
T cells, IL-17A, and psoriasis
Since the demonstrated efficacy of cyclosporine in psoriasis (4), alpha beta T cells have been thought to be central to psoriasis pathophysiology, and recent clinical evidence points to the Th17 subtype as the driver of disease (5), as is evident by the success of IL-17/IL-23-targeted therapies (6-9). However, many groups are pursuing other sources of IL-17 as important mediators of the psoriatic phenotype. For example, some investigators have posited that in psoriasis, gamma delta T cells are expanded disproportionately more than alpha beta T cells, and that the former is the dominant source of IL-17 (10-12).
To address the debate of alpha beta versus gamma delta T cells head on we compared the relative frequencies of the corresponding alpha, beta, gamma, delta gene segments in psoriatic and normal skin (surprisingly nobody has done this before). By looking at relative gene expression, we found no evidence for a differential expansion of gamma delta T cells over alpha beta T cells. In fact, gamma delta TCR gene segments were only marginally over expressed in the setting of psoriasis. We next sought to address the source of IL-17 by plotting the expression of individual TCR gene segments against IL17A expression. Again, we found no evidence that gamma delta TCR gene segments correlated with IL17A expression. Interestingly, TRGV5 expression did correlate with IL36. In contrast, TRAJ23 expression correlated well with IL17A. These results were then validated in four independently collected RNA-Seq datasets. From these results we speculate that TRAJ23-expressing cells (abundant in psoriasis) are a source of IL-17 and TRGV5-expressing T cells possibly expand in response to IL-36, a psoriasis-associated cytokine. These results are summarized in the figure below. For more details of this study and to see our other exciting discoveries please see the published manuscript by clicking here.
1. Chang YT, Liu HN, Shiao YM, Lin MW, Lee DD, Liu MT, et al. A study of PSORS1C1 gene polymorphisms in Chinese patients with psoriasis. The British journal of dermatology. 2005;153(1):90-6.
2. Menssen A, Trommler P, Vollmer S, Schendel D, Albert E, Gurtler L, et al. Evidence for an antigen-specific cellular immune response in skin lesions of patients with psoriasis vulgaris. Journal of immunology. 1995;155(8):4078-83.
3. Vollmer S, Menssen A, and Prinz JC. Dominant lesional T cell receptor rearrangements persist in relapsing psoriasis but are absent from nonlesional skin: evidence for a stable antigen-specific pathogenic T cell response in psoriasis vulgaris. The Journal of investigative dermatology. 2001;117(5):1296-301.
4. Ellis CN, Fradin MS, Messana JM, Brown MD, Siegel MT, Hartley AH, et al. Cyclosporine for plaque-type psoriasis. Results of a multidose, double-blind trial. N Engl J Med. 1991;324(5):277-84.
5. Thaci D, Blauvelt A, Reich K, Tsai TF, Vanaclocha F, Kingo K, et al. Secukinumab is superior to ustekinumab in clearing skin of subjects with moderate to severe plaque psoriasis: CLEAR, a randomized controlled trial. J Am Acad Dermatol. 2015;73(3):400-9.
6. Langley RG, Elewski BE, Lebwohl M, Reich K, Griffiths CE, Papp K, et al. Secukinumab in plaque psoriasis--results of two phase 3 trials. N Engl J Med. 2014;371(4):326-38.
7. McInnes IB, Mease PJ, Kirkham B, Kavanaugh A, Ritchlin CT, Rahman P, et al. Secukinumab, a human anti-interleukin-17A monoclonal antibody, in patients with psoriatic arthritis (FUTURE 2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2015;386(9999):1137-46.
8. Papp KA, Leonardi C, Menter A, Ortonne JP, Krueger JG, Kricorian G, et al. Brodalumab, an anti-interleukin-17-receptor antibody for psoriasis. N Engl J Med. 2012;366(13):1181-9.
9. Gordon KB, Blauvelt A, Papp KA, Langley RG, Luger T, Ohtsuki M, et al. Phase 3 Trials of Ixekizumab in Moderate-to-Severe Plaque Psoriasis. N Engl J Med. 2016;375(4):345-56.
10. Cai Y, Shen X, Ding C, Qi C, Li K, Li X, et al. Pivotal role of dermal IL-17-producing gammadelta T cells in skin inflammation. Immunity. 2011;35(4):596-610.
11. Yoshiki R, Kabashima K, Honda T, Nakamizo S, Sawada Y, Sugita K, et al. IL-23 from Langerhans cells is required for the development of imiquimod-induced psoriasis-like dermatitis by induction of IL-17A-producing gammadelta T cells. J Invest Dermatol. 2014;134(7):1912-21.
12. Hartwig T, Pantelyushin S, Croxford AL, Kulig P, and Becher B. Dermal IL-17-producing gammadelta T cells establish long-lived memory in the skin. Eur J Immunol. 2015;45(11):3022-33.


My Profound and Utmost Gratitude to Doctor Razor, I am here to Share my testimony about a doctor who helped me remove a burden from my life. I was diagnosed with HSV 1 on March 17, 2017, i didn't bother seeking for medical aide because i know that the Government And medical Scientists Have been lying to us about herpes not Having a complete Cure, so I was thinking of how to get a solution, To make sure i get this Infection out of my body. I went to visiting a friend of mine who happens to be A family friend as well and she asked why i was looking so sad, i explained to her my problem, she told me that she can help me out, she introduced me to a doctor who uses herbal medication to cure HERPES SIMPLEX VIRUS and gave me his email, as well as his Website : https://drrazorherbalhome.wixsite.com/drrazorherbalhome . So I emailed him. He told me all the things I needed to do and also gave me instructions on How i was to take the medicine For 18 days, which I followed properly. After completion of the Dosage the herbalist Requested that i go for a medical Test And i was amazed after my Test Result came out Negative. If you are also heart broken and also need help, you can also email him at: drrazorherbalhome@gmail.com . Whatsapp/Call Doctor Razor via +2349065420442
ReplyDeleteHello everyone, my name is Alfreed siang i am here to say a big thank you to my doctor DR OLU who helped me enlarge my penis.i have never had a happy relationship in my life because of my inability to perform well due to my small penis, due to frustration,i went online in search of solution to ending my predicament and than i came across testimony on how DR OLU has helped them, so i contacted him and he promised to help me with penis enlargement,i doubted at first but i gave him a trial and he sent me the product which i used according to his prescription and in less than a week,i saw changes in my penis and it grow to the size i wanted and since then,i am now a happy man and no lady complains again about my penis.if you also need the services of my doctor,you can also contact him on his email.. drolusolutionhome@gmail.com or his whataspp is +2348140654426
DeleteHello everyone, my name is Alfreed siang i am here to say a big thank you to my doctor DR OLU who helped me enlarge my penis.i have never had a happy relationship in my life because of my inability to perform well due to my small penis, due to frustration,i went online in search of solution to ending my predicament and than i came across testimony on how DR OLU has helped them, so i contacted him and he promised to help me with penis enlargement,i doubted at first but i gave him a trial and he sent me the product which i used according to his prescription and in less than a week,i saw changes in my penis and it grow to the size i wanted and since then,i am now a happy man and no lady complains again about my penis.if you also need the services of my doctor,you can also contact him on his email.. drolusolutionhome@gmail.com or his whataspp is +2348140654426
I've Used Dr Aziba Product to solve my Penis Growth problem of 3.8 inches to 9.0 Inches in Less than Seven Days.
DeleteTwo hours after starting the Product, I began to feel its results. My penis was tingling (but it did not hurt), and little by little it was growing. Three days later, it already measured 6.6 inches. I had to do absolutely nothing because it was completely automatic. Six days later, I had gained one more Eight inch. The result: 8.1 inches! I began to feel more confident as I slept with my Girlfriend, who is always willing to do the dirty. In the end, she told me a lot has changed in me and i now gives her so much orgasms in half an hour. i'm recommending you with similar problem to Get In Contact with Doctor Aziba via Email: Priestazibasolutioncenter@gmail.com & WhatsApp (+234)810-0368-288 For Help!
I've Used Dr Aziba Product to solve my Penis Growth problem of 3.8 inches to 9.0 Inches in Less than Seven Days.
Two hours after starting the Product, I began to feel its results. My penis was tingling (but it did not hurt), and little by little it was growing. Three days later, it already measured 6.6 inches. I had to do absolutely nothing because it was completely automatic. Six days later, I had gained one more Eight inch. The result: 8.1 inches! I began to feel more confident as I slept with my Girlfriend, who is always willing to do the dirty. In the end, she told me a lot has changed in me and i now gives her so much orgasms in half an hour. i'm recommending you with similar problem to Get In Contact with Doctor Aziba via Email: Priestazibasolutioncenter@gmail.com & WhatsApp (+234)810-0368-288 For Help!
I have been suffering from (HERPES) disease for the past 10 years and had constant pain, especially in my knees. During the 4th month , I had faith in God that
DeleteI would be healed someday.The pain started to circulate all over my body and I have been taking treatment from my doctor, 5 months ago I came on search on
the internet if I could get any information concerning the prevention of this disease, on my search I saw a testimony of someone who has been healed from
(Hepatitis B and Cancer) by this Man DR festus with the address of this man and advise we should contact him for any sickness that he would be of help, so I
wrote to DR festus telling him about my HERPES Virus and chronic pain all over my body and he told me not to worry that I was going to be cured!! hmm i never
believed it either,, well after all the procedures and remedy given to me by this man few weeks later I started experiencing changes all over me as the DR
assured me that I have been cured with the chronic pain gone after the first week of using his medicine, after 2 weeks i went to my doctor to confirmed if
I have been finally healed and the result came out negative, So friends my advice is if you have such sickness or any other like HSV1,2,HPV1,2,3,CANSER HIV,COLD
SORES,TOOTH ache, ETC you can email DR Festus on drfestusherbalsolutioncentre@gmail.com sir I am indeed grateful for the help I will forever recommend you to my
friends!!! you can also text him on his whatsapp +2347073677389.
Are you in need of finance? we give out guarantee cash at 3% interest rate. Contact us on any kind of finance now: financialserviceoffer876@gmail.com whatspp Number +918929509036 Dr James Eric
ReplyDelete
DeleteWords are empty when spoken. A big thank you to Dr Aziba for using his effective herbal product to reserve my ALS disease. I was diagnosed of ALS disease a year back. My symptoms progressed so very fast that i couldn't do anything. I had tripping, dropping, abnormal fatigue of my leg, slurred speech, muscle cramps and twitches. I was very depressed and had no hope of living. i couldn't resist it than to go deep into research for a cure. On this fateful day, i saw a testimony post concerning a lady who once had ALS disease but was reserved using Dr Aziba Natura Herbal product. At this time, i was skeptical and curious. To cut it short, i visited his WhatsApp on +2348100368288 and i saw so many review which motivated me to make my orders. I received the product and used it as instructed for two weeks. Omg, during the second week of usage, all my symptoms disappeared, today makes it 3 months after usage and i can boldly tell to the public that i have been reserved from ALS disease for 3 years span and after which i will make another orders again after 3 years.. This is more than a miracle, i just can't believe my life is back again. last 2 months i resumed work and i can now do all activities myself without anyone's assistance. You that is out there searching for solution, i have bring good news to you kindly Contact Dr via Email. Priestazibasolutioncenter@gmail.com or reach him directly via WhatsApp +2348100368288 Today marks my birthday. Please lets share this to help others. Thank you Dr Aziba for your Herbal product.
NATURAL HERPES CURE.Herpes simplex virus is said to have no cure, just as i was told by my family doctor that herpes can not be cured but can be controlled, Although i was a very stubborn person i never believed in what my doctor told me about herpes having no cure. Am a very honest person who believe in nature and i also believed that there could be cure somewhere in the world, I kept my faith so strong and kept on doing my research till i saw an article shared by a testifier on How a herbal Doctor Razor from west Africa, i Decided to use his herbal medicine, so i Reached out to him and explained my situation to him, he promised to guide me, after 4 days of reaching out to him i received my medication sent to me by Doctor Razor and he instructed me to use it for 18 days, After Completion of his herbal medicine dosage, i went for a medical checkup as i was told by Doctor Razor. I am so happy and extremely excited that I got cured of this Virus. Massive gratitude to doctor Razor. Reach out to him via his clinic email : drrazorherbalhome@gmail.com On whatsapp/call his cell phone +2349065420442. Together we stand a chance to fight herpes. Let's continue to share this till The whole world Gets to know that there is a herbal cure for herpes.God bless you all.
ReplyDeleteI was able to get rid of my herpes virus this year, i was diagnosed of herpes virus since late 2015 and i was on my doctor prescription ever since i have the virus, I wasn’t satisfied i needed to get the virus out of my system and get rid of the virus,i was going through a blog to find new informations about the Herpes Simplex Virus i came across a review of a lady who was testify on the cured that the great herbalist cured is virus and i have to contert him and see what he told me to do for me to get rid of the cure I contacted the herbal foundation and they guided me on how to purchase the herbal formula. the herbal products las me for 3 weeks which helps me to get rid of the virus totally, for easy communication you can reach Dr Freeman Osas on his WhatSapp via +2347057052206 email. balogospellcaster@gmail.com
ReplyDeleteI was able to get rid of my herpes virus this year, i was diagnosed of herpes virus since late 2015 and i was on my doctor prescription ever since i have the virus, I wasn’t satisfied i needed to get the virus out of my system and get rid of the virus,i was going through a blog to find new informations about the Herpes Simplex Virus i came across a review of a lady who was testify on the cured that the great herbalist cured is virus and i have to contert him and see what he told me to do for me to get rid of the cure I contacted the herbal foundation and they guided me on how to purchase the herbal formula. the herbal products las me for 3 weeks which helps me to get rid of the virus totally, for easy communication you can reach Dr Freeman Osas on his WhatSapp via +2347057052206 email. balogospellcaster@gmail.com
ReplyDeleteMy ex-husband and I had always managed to stay friendly after our divorce in February 2017. But I always wanted to get back together with him, All it took was a visit to this spell casters website last December, because my dream was to start a new year with my husband, and live happily with him.. This spell caster requested a specific love spell for me and my husband, and I accepted it. And this powerful spell caster began to work his magic. And 48 hours after this spell caster worked for me, my husband called me back for us to be together again, and he was remorseful for all his wrong deeds. My spell is working because guess what: My “husband” is back and we are making preparations on how to go to court and withdraw our divorce papers ASAP. This is nothing short of a miracle. Thank you Dr Emu for your powerful spells. Words are not enough. here is his Email: emutemple@gmail.com or call/text him on his WhatsApp +2347012841542
ReplyDeleteHey �� everyone. I just wanna tell y’all that I’ve been cured from herpes virus. It only took me Two weeks to be cured finally from this virus and what I did definitely got me cured completely from my HSV2, I’m so happy ������. When every odd seems to be against me i trusted in the lord. I battled for so long until a friend introduced me to Doctor Kham who was finally able to help me and here i am today healthy and strong. Email this herbalist via dr.khamcaregiver@gmail.com that is his contact details.
ReplyDelete
ReplyDeleteWords are empty when spoken. A big thank you to Dr Aziba for using his effective herbal product to reserve my ALS disease. I was diagnosed of ALS disease a year back. My symptoms progressed so very fast that i couldn't do anything. I had tripping, dropping, abnormal fatigue of my leg, slurred speech, muscle cramps and twitches. I was very depressed and had no hope of living. i couldn't resist it than to go deep into research for a cure. On this fateful day, i saw a testimony post concerning a lady who once had ALS disease but was reserved using Dr Aziba Natura Herbal product. At this time, i was skeptical and curious. To cut it short, i visited his WhatsApp on +2348100368288 and i saw so many review which motivated me to make my orders. I received the product and used it as instructed for two weeks. Omg, during the second week of usage, all my symptoms disappeared, today makes it 3 months after usage and i can boldly tell to the public that i have been reserved from ALS disease for 3 years span and after which i will make another orders again after 3 years.. This is more than a miracle, i just can't believe my life is back again. last 2 months i resumed work and i can now do all activities myself without anyone's assistance. You that is out there searching for solution, i have bring good news to you kindly Contact Dr via Email. Priestazibasolutioncenter@gmail.com or reach him directly via WhatsApp +2348100368288 Today marks my birthday. Please lets share this to help others. Thank you Dr Aziba for your Herbal product.
HERBAL DR EMU WHO PREPARE HERBAL MEDICINE TO CURE ALL KINDS OF DISEASES INCLUDING HERPES DISEASE.
ReplyDeleteI have been battling this Herpes disease for almost 3 years now....I tried all possible means to get cure from my Herpes Disease but all to be in vain until i saw a post in a health forum about a herbal Dr Emu who prepare herbal medicine to cure all kind of diseases including Herpes Disease, at first i doubted if it was real but decided to give it a try...when i contact Dr Emu via his email (emutemple@gmail.com) write him and reply me explain how the process work so after ordering for the medicine I got it within 3/4 working days through DHL Delivery and I took it according to the way Dr Emu instructed, I was so happy after 2 week I took the medicine there was very big change in my health when I was done with the process I go for test, I found out I am negative...Herpes patients should also get in touch with this herbalist Dr Emu to get rid of these Herpes Virus forever his whatsapp number +2347012841542.
My name is Brittney Brian,i am from Arizona, Its a pleasure for me to write this testimony about how i got my Genital Herpes cured 2 months ago. i have been reading so many comments of some people who were cured from various diseases by Dr. Festus, I was hurt and depressed so I was too curious and wanted to try Dr. Festus cuz have been burdened by this virus for many , then i contacted him through his email when i contact him, he assured me 100% that he will heal me, i pleaded with him to help me out which he did, he healed me just as he promised, he sent me his medication and ask me to go for check up after 5 weeks of taking the medication. i agreed with him i took this medication and went for check up, to my greatest surprise my result was negative after the treatment, i am really happy that i am cured and healthy again. I have waited for 5 weeks after I first tested negative just to be very sure i was completely healed before writing this testimony I just got back from the hospital today from getting my blood test result and am still negative, so i guess its time i recommend anyone going through Herpes HSV-1 or HSV-2, HIV, HPV, Hepatitis A&B, Diabetes, Cancer reach him through Email drfestusherbalsolutioncentre@gmail.com OR add on whatsapp +2348073742265.
ReplyDeleteI've Used Dr Aziba Product to solve my Penis Growth problem of 3.8 inches to 9.0 Inches in Less than Seven Days.
ReplyDeleteTwo hours after starting the Product, I began to feel its results. My penis was tingling (but it did not hurt), and little by little it was growing. Three days later, it already measured 6.6 inches. I had to do absolutely nothing because it was completely automatic. Six days later, I had gained one more Eight inch. The result: 8.1 inches! I began to feel more confident as I slept with my Girlfriend, who is always willing to do the dirty. In the end, she told me a lot has changed in me and i now gives her so much orgasms in half an hour. i'm recommending you with similar problem to Get In Contact with Doctor Aziba via Email: Priestazibasolutioncenter@gmail.com & WhatsApp (+234)810-0368-288 For Help!
I've Used Dr Aziba Product to solve my Penis Growth problem of 3.8 inches to 9.0 Inches in Less than Seven Days.
ReplyDeleteTwo hours after starting the Product, I began to feel its results. My penis was tingling (but it did not hurt), and little by little it was growing. Three days later, it already measured 6.6 inches. I had to do absolutely nothing because it was completely automatic. Six days later, I had gained one more Eight inch. The result: 8.1 inches! I began to feel more confident as I slept with my Girlfriend, who is always willing to do the dirty. In the end, she told me a lot has changed in me and i now gives her so much orgasms in half an hour. i'm recommending you with similar problem to Get In Contact with Doctor Aziba via Email: Priestazibasolutioncenter@gmail.com & WhatsApp (+234)810-0368-288 For Help!
This is very helpful with no side effect...Before I began Doctor Aziba natural penis enlargement Product I measured 4 Inches in erect length, and 3 inches in erect girth. While flaccid I measured 3.4 Inches long. I began using his product, slightly skeptical I admit, and just 2 weeks later I was very happy with my progress. I measured 7.5 inches erect length and 6.5 inches erect girth, and 5.5 inches flaccid length. I was actually quite shocked at the huge gains I’d had, and continued using the product regularly, sticking to the usage schedule rigorously. After about 2-3 weeks I am 8.2 Inches in erect length and 8.1 Inches in erect girth. I am also 7.2 fnches long while flaccid. This has had a huge impact on my life, not only has my sex life improved greatly, I feel much more confident and proud. Dr Aziba Natural Penis Enlargement works! and you can as well reach out to him on any of these cure for ALS, HPV, HSV, HIV, HERPES, Virginal Infection, Heart Burn, Cancer etc... You can Get in Contact with his via Email: (Priestazibasolutioncenter@gmail.com ) or WhatsApp him via contact +2348100368288 His the solution you've been searching !
ReplyDeleteThis is very helpful with no side effect...Before I began Doctor Aziba natural penis enlargement Product I measured 4 Inches in erect length, and 3 inches in erect girth. While flaccid I measured 3.4 Inches long. I began using his product, slightly skeptical I admit, and just 2 weeks later I was very happy with my progress. I measured 7.5 inches erect length and 6.5 inches erect girth, and 5.5 inches flaccid length. I was actually quite shocked at the huge gains I’d had, and continued using the product regularly, sticking to the usage schedule rigorously. After about 2-3 weeks I am 8.2 Inches in erect length and 8.1 Inches in erect girth. I am also 7.2 fnches long while flaccid. This has had a huge impact on my life, not only has my sex life improved greatly, I feel much more confident and proud. Dr Aziba Natural Penis Enlargement works! and you can as well reach out to him on any of these cure for ALS, HPV, HSV, HIV, HERPES, Virginal Infection, Heart Burn, Cancer etc... You can Get in Contact with his via Email: (Priestazibasolutioncenter@gmail.com ) or WhatsApp him via contact +2348100368288 His the solution you've been searching !
Hello everybody
ReplyDeletei don't just know the reason why some people is finding it difficult to believe that there is a cure for herpes, i have been suffering from herpes since last three years with my boyfriend but today i am happy that am cure from it with the herbal medicine of Dr eromonsele the great healer,i was browsing the internet searching for help when i came across a testimony shared by someone on how Dr eromonsele cure his herpes i was so much in need of getting his treatment but after all Dr eromonsele brought a smile to my face with his herbal medicine. i am so much happy today that we have someone like this great healer out there, so my people out there kindly contact this great healer on his email address: dr.eromonsele@gmail.com please sir keep your good work cause there are people out there who is in need of your healing medicine.once more contact him now: dr.eromonsele@gmail.com you can call him or whatsApp his number +2349079308479
Contact Dr DADA for penis enlargement pills and premature ejaculation and week erection,disfution ,diabetes, Cancer, heart problem, breast enlargement and hip enlargement. Dr DADA help me with penis enlargement pills so if you are in the same position like me contact him ( drdadaspellhome7@gmail.com ) OR whatsapp him on +2349023126215
ReplyDeleteAm very glad to share this testimony with everyone for the marvelous work Dr DADA has done for my life, 6months ago i was diagnosed with herpes virus and ever since then i have been very unhappy, i was so down broken everyday, until one day when i came across a shocking testimony about how Dr DADA cured someone. of his herpes virus, without wasting much time i contacted him immediately on his email address: < drdadaspellhome7@gmail.com and after i explain myself to him about how terrible i have been, and he assure me that he will help me to cure my herpes virus,after he has prepared the herbal medicine, he sent it to me and when i have received it and started using it i was totally cure within 2weeks, i am forever grateful to Dr DADA for helping me out with his herpes prescription that cured my virus. contact his email addreSS (drdadaspellhome7@gmail.com or you can call or whatsApp his Mobile number:2349023126215
ReplyDeleteThis comment has been removed by the author.
ReplyDeleteThis comment has been removed by the author.
ReplyDeleteSince it needs first financial investment; which is extremely marginal contrasted to the advantages of ease as well as even more exact outcomes, this approach is no longer complimentary. You desire to carry out the complimentary criminal document pascocountyarrests check and also you do not desire to invest a solitary dollar on your lookup; is this truly feasible?
ReplyDeleteQuick Cure in Eradicating Herpes Simplex Virus by Doctor ELINFOH. I recommend doctor Elinfoh herbal medicine to anyone battling Herpes Infection...I am so happy to be finally cured of herpes simplex virus by herbalist Elinfoh,Ever since I was diagnosed with the virus on June 24, 2018. I have been using medical drugs to suppress it, I later realized it was causing more damage to my health than the virus,I opted for herbs and got to find out about the great work herbalist Doctor Elinfoh has been doing with his herbal remedy. I contacted him and he promised to help me get rid of it,after 18 days I received a package from the UPS delivery service and it was from the doctor who sent me the herbal treatment in Dallas,Texas USA, And he instructed me on how I was going to take it for the next 3 weeks . After completing the dosage he told me to go for a medical checkup and when I did the doctors conducted some tests on me but they were stunned that my status was now negative,they conducted several tests on me to be sure. I even went to Medical City Dallas Hospital, where they conducted another test and it was still negative. I am so grateful to you herbal doctor Elinfoh , God will continue to prosper you,I pray the world gets to know about you and your herbal exploits. He also cures any other Human diseases. Reach him on his email drelinfohherbalhome@gmail.com PLEASE SHARE , SO THE WORLD CAN GET TO KNOW ABOUT HIM.DR ELINFOH HAS THE BEST HERBAL REMEDY FOR HERPES SIMPLEX VIRUS 1&2.
ReplyDeleteDR EMU WHO HELP PEOPLE IN ANY TYPE OF LOTTERY NUMBERS
ReplyDeleteIt is a very hard situation when playing the lottery and never won, or keep winning low fund not up to 100 bucks, i have been a victim of such a tough life, the biggest fund i have ever won was 100 bucks, and i have been playing lottery for almost 12 years now, things suddenly change the moment i came across a secret online, a testimony of a spell caster called dr emu, who help people in any type of lottery numbers, i was not easily convinced, but i decided to give try, now i am a proud lottery winner with the help of dr emu, i won $1,000.0000.00 and i am making this known to every one out there who have been trying all day to win the lottery, believe me this is the only way to win the lottery.
Dr Emu can also help you fix this issues
(1)Ex back.
(2)Herbal cure & Spiritual healing.
(3)You want to be promoted in your office.
(4)Pregnancy spell.
(5)Win a court case.
Contact him on email Emutemple@gmail.com
What’s app +2347012841542
Website Https://emutemple.wordpress.com/
Facebook page Https://web.facebook.com/Emu-Temple-104891335203341
MY HIV HEALING TESTIMONY
ReplyDeleteMy mouth is short of words, i am so happy because Dr voodoo has healed me
from HIV which i have been suffering from the past 5years now, i have
spend a lot when getting drugs from the hospital to keep me healthy, i have
tried all means in life to always i can become HIV negative one day, but
there was no answer until i found Dr voodoo the parish of African who
provide me some healing herbs that i uses , now i am glad telling everyone
that i am now HIV Negative, i am very happy, thank you Dr voodoo for
helping my life comes back newly without any form of crisis, may the good
lord that i serve blessed you Dr voodoo and equip you to the higher grade
for healing my life. i am so amazed. so i will announced to everyone in
this whole world that is HIV positive to please follow my advice and get
healed on time, because we all knows that HIV disease is a deadly
type,contact Dr voodoo for your HIV healing herbs today at:
voodoospelltemple66@gmail.com He will be always happy to assist you
online and ensure you get healed on time, contact Dr voodoo today for
your healing hearbs immediately, you can also
add him on whatsApp +2348140120719 thank you sir.
I have read so much on Herpes Simplex virus. I hear of reaching clearance, I hear of no cure, I hear of recurrent lesions, poor immune system, poor diet, etc. I just do not know what to believe. I have visited many web pages that seem to sell you their version of removing or enforcing the immune system. Then I hear all the info on therapeutic vaccines as well as prophylactic vaccines and wonder how that can even help me. And I feel so worthless, Glad I came across a Doctor Razor article on how to heal HSV forever. Endless Gratitude to doctor Razor for helping me get rid of my Herpes Virus. Reach him on his clinic email drrazorherbalhome@gmail.com
ReplyDeleteMessage Herbalist Razor on WhatsApp. https://wa.me/message/USI4SETUUEW4H1
Life is always beautiful when you have good health. I have been suffering from Herpes and I was looking for help in every ways and luckily I was directed to a very kind and Great DR HARRY HERBS, who helped me cure my Herpes and today I am free from Herpes and very healthy thank you so much DR HARRY HERBS. kindly reach him and he's helpful.
ReplyDeletewebsite doctorharryherbs.com
email Drharryherbspell01@gmail.com
I have been suffering from (HERPES) disease for the past 10 years and had constant pain, especially in my knees. During the 4th month , I had faith in God that
ReplyDeleteI would be healed someday.The pain started to circulate all over my body and I have been taking treatment from my doctor, 5 months ago I came on search on
the internet if I could get any information concerning the prevention of this disease, on my search I saw a testimony of someone who has been healed from
(Hepatitis B and Cancer) by this Man DR festus with the address of this man and advise we should contact him for any sickness that he would be of help, so I
wrote to DR festus telling him about my HERPES Virus and chronic pain all over my body and he told me not to worry that I was going to be cured!! hmm i never
believed it either,, well after all the procedures and remedy given to me by this man few weeks later I started experiencing changes all over me as the DR
assured me that I have been cured with the chronic pain gone after the first week of using his medicine, after 2 weeks i went to my doctor to confirmed if
I have been finally healed and the result came out negative, So friends my advice is if you have such sickness or any other like HSV1,2,HPV1,2,3,CANSER HIV,COLD
SORES,TOOTH ache, ETC you can email DR Festus on drfestusherbalsolutioncentre@gmail.com sir I am indeed grateful for the help I will forever recommend you to my
friends!!! you can also text him on his whatsapp +2347073677389.
I will forever be grateful for the miraculous work you did for me Dr Ogudugu, words are not enough to express my gratitude. I was diagnosed of herpes in 2022 and I tried all possible means to get cure but all to no avail until I saw a post in a health forum about a herbalist who prepares herbal medicine to cure all kind of diseases including herpes virus, at first i doubted if it was real but decided to give it a try, when i contacted this herbal doctor via his email i bought the herpes herbal medicine and received it through DHL within 5 working days and used it as prescribed, i tested negative to herpes virus within 14 days of use. Thanks to Dr Ogudugu His email: greatogudugu@gmail.com And WhatsApp Number +2348163026536.
ReplyDelete